An estimated 10% of adults in the United States are affected by hyperactive inflammation of the skin called eczema (detail explanation) which causes rashes, dryness and itchiness in mild condition to blistering and bleeding of the skin in extreme cases. This type of skin disease is caused by a mixture of genetic and environmental factors like extreme temperature and dust particles in the wind.
The application of steroids as a treatment for patients suffering from eczema is the most common dermatological practice, but this kind of treatment suppresses the body’s inflammatory response thereby weakening our immune system. Aside from this, Dermatologist suggests that treatment using steroids are not safe due to side effects like weakening of the skin, kidney damage and osteoporosis.
Thanks to modern research as scientist were able to isolate and identify the molecular protein found in our body as the triggering mechanism for eczema. This discovery has led to the development of new treatment drugs and has changed dermatologists look at eczema as it has previously linked entirely to the weakening of the immune system.
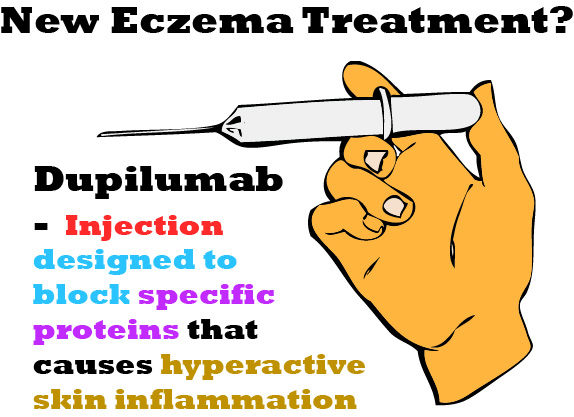
Injection Treatment – Dupilumab
Dupilumab, an injectable developed by Regeneron Pharmaceuticals in partnership with Sanofi, is a treatment drug designed to block specific proteins that causes hyperactive skin inflammation. Its initial success during clinical trials has earned its designation from the US Food and Drug Administration (FDA) as a “Breakthrough Therapy” where the patients’ skin condition was lowered down from extreme rashes covering almost the entire body to having a perfectly natural skin tone.
Ointment Application Treatment – Crisborole
The previous treatment for patients with mild eczema uses a steroidal pill that blocks a particular enzyme known as PDE-4 which is known to cause skin inflammation. However, the side effects of taking steroidal pills like nausea and diarrhea is considered too harsh for patients only suffering from a mild condition.
Crisaborole, an ointment developed by Anacor Pharmaceuticals, is a new treatment that can be applied directly to the skin without experiencing serious side effects since the drug will not circulate throughout the entire body system. This drug employs the principle of topical skin medication and with the efficiency of Boron, as one of its ingredients, can better penetrate the skin and hits the target molecule PDE-4 thereby stopping inflammation.
Still Needs More Studies
Clinical studies show that the simultaneous treatment of eczema using the injectable dupilumab and the topical application of crisaborole ointment facilitates fast recovery. Patients who participated during clinical trials claimed that it is not just the debilitating rashes that have been cured, but also regained self-esteem and confidence. Patients said they are afraid to go out as some of their friends refuses to have direct contact with them bearing in their minds that this type of disease is contagious. It is not.
Patient is needed as both of these treatments are still on the final stage of approval and the US-FDA is yet to decide whether these drugs will become the first non-steroidal treatment for eczema. If all goes well, dupilimab and crisaborole will be out in the market on 2017.










Helllo!! My name is Sou
I have lived with Eczema my whole life and it has been a constant battle. Welcome to my journey! Let's battle this together!